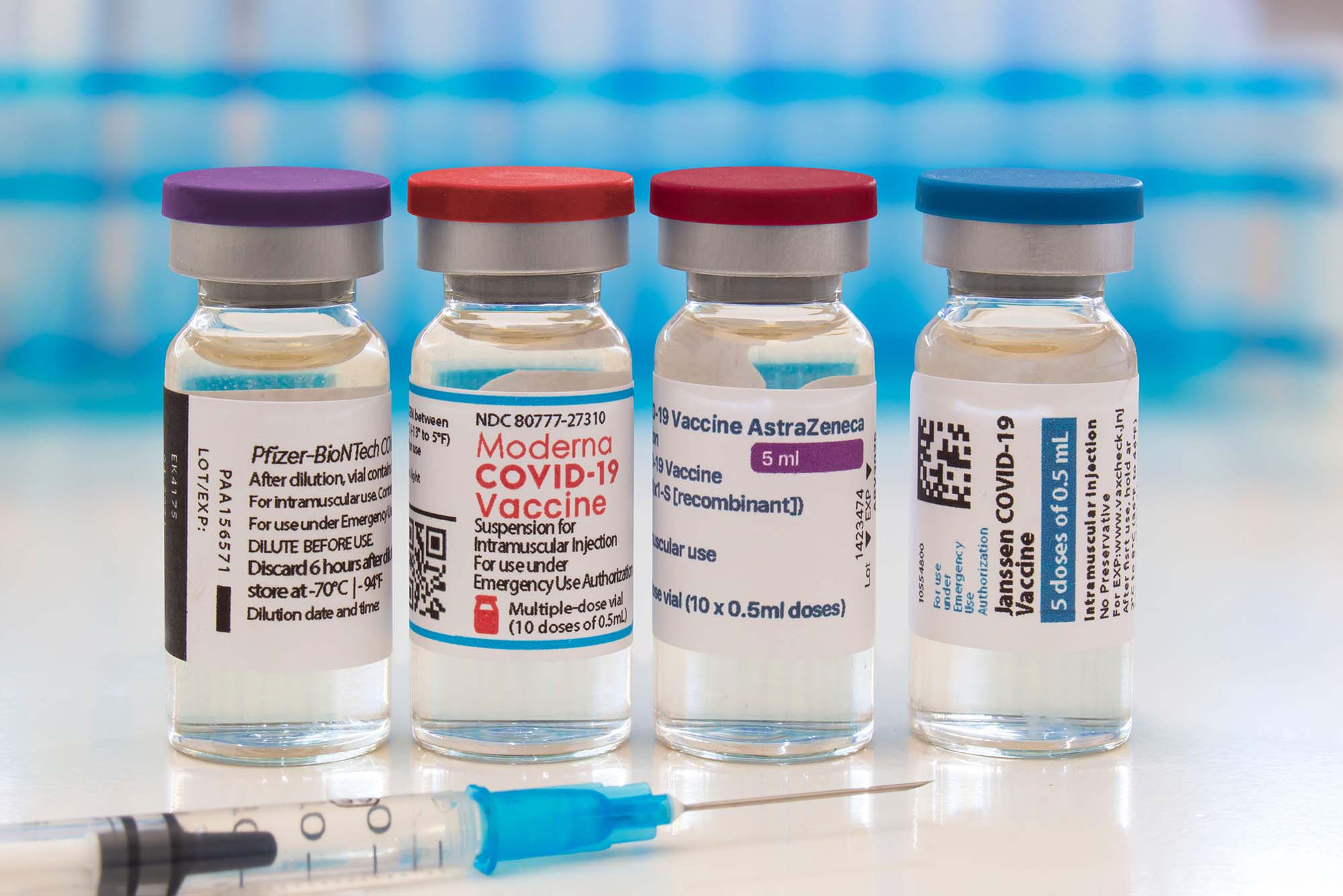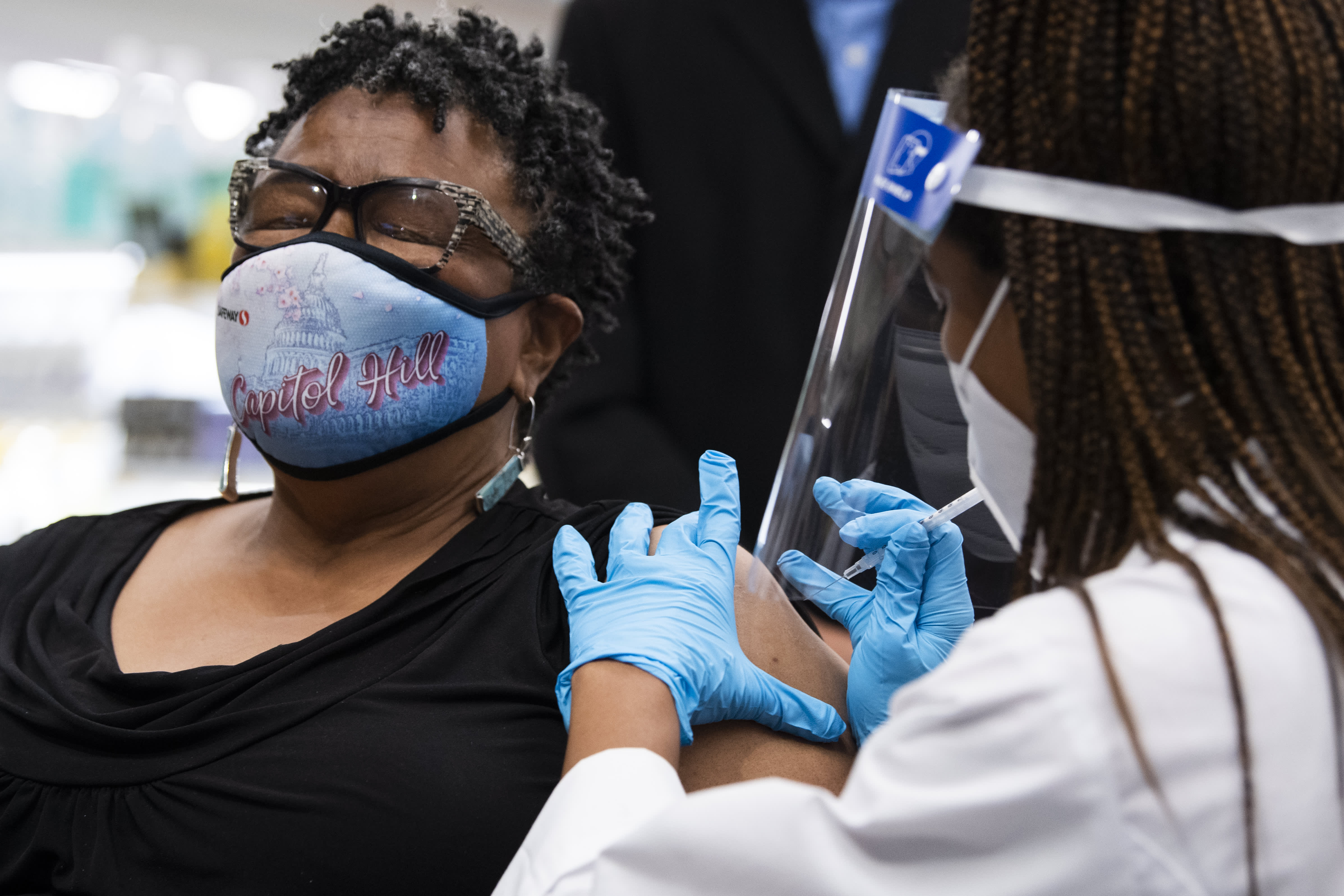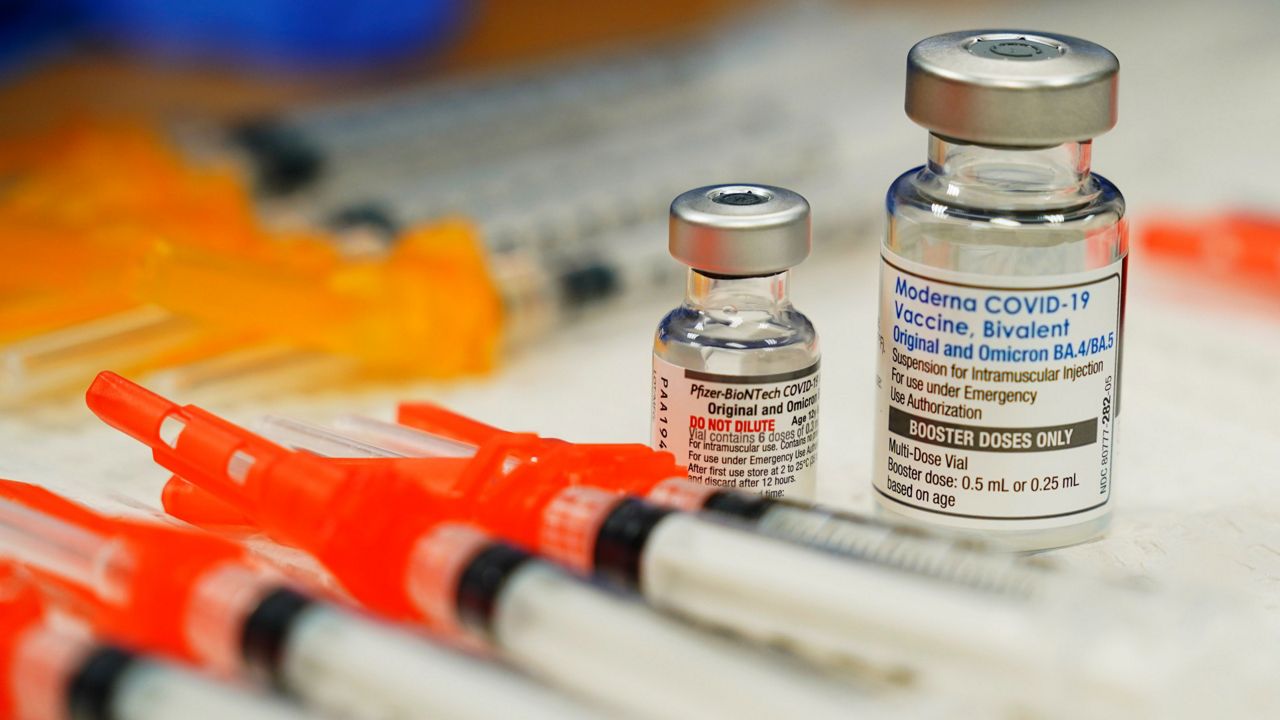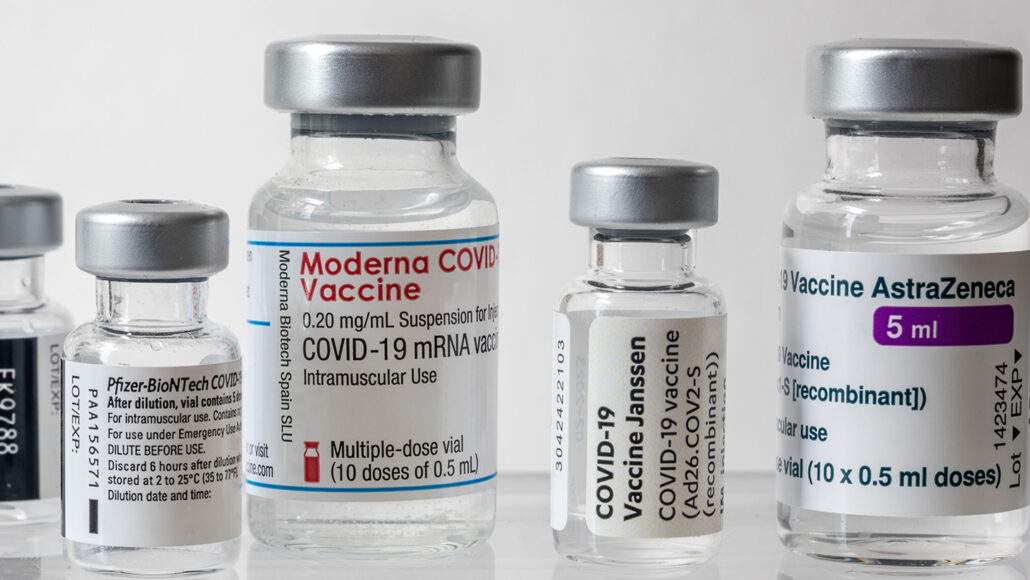
LVHN Prepared to Give Pfizer COVID-19 Vaccine Booster Shot to Seniors, At-Risk Workers, Vulnerable Populations Starting Sept. 27
Garantizar la vacunación en colectivos en riesgo: el reto europeo en el tercer invierno de pandemia | Euronews
Natividad Hosting Pfizer-BioNTech COVID-19 Vaccine and Booster Clinics Every Wednesday from 2:00 to 4:00 pm - Natividad: Inspiring healthy lives

Lawrence General Hospital - The Pfizer-BioNTech COVID-19 vaccine booster has been approved by the US Food and Drug Administration. You may schedule your appointment at https://bit.ly/3iAVUhn Eligibility is: • MUST have received

Pfizer-BioNTech will seek authorization for second COVID booster for older adults | KPBS Public Media